How AI agents work across the regulatory lifecycle

AI Agents
AI Agents
AI Agents
By Sofía Sánchez González
The first time you heard the term AI agent, you probably pictured a tiny robot in a sharp uniform, maybe carrying a clipboard and reporting for duty. Or at best, an upgraded chatbot that politely answers questions and calls it a day.
But as you have probably realized by now, AI agents are a completely different species: They do not have a surveillance role and they do not “just” generate text. They plan, retrieve information, interact with tools, and execute multi-step tasks inside real workflows.
And especially in the pharmaceutical sector, that distinction is critical. Clinical development, regulatory affairs, and manufacturing are not one-step processes. They involve data, validation, compliance, and structured decision-making. That is where AI agents make the difference.
So, what exactly is an AI agent in pharma, and how does it actually work?
What is an AI agent, really?
Before we dive into applications, we need to clarify one thing: an AI agent is not just a model. As we explained in a previous post, it’s a system built around a model. A traditional AI model, just like ChatGPT, Gemini, or Claude, works like this:
- You ask a question.
- It generates a response.
- The interaction ends.
An AI agent works differently:
- You define a goal.
- It breaks that goal into steps.
- It decides what data it needs.
- It calls the right tools.
- It executes actions in sequence.
- It evaluates the outcome.
Instead of reacting, it orchestrates. That orchestration layer is what turns AI into an operational system.
Reactive AI vs agentic AI: a simple comparison
All of that theory sounds great, but it might be easier to understand with a concrete example from the life sciences sector. Let’s imagine a real-world pharma scenario. You ask:
“Draft the efficacy section of the Clinical Study Report and ensure alignment with the statistical analysis plan.”
A standard generative model would:
- Produce a draft based on the text you provide.
- Summarize tables if they are pasted into the prompt.
- Generate generic regulatory language.
An AI agent-based solution would:
- Retrieve the protocol and statistical analysis plan.
- Extract relevant tables, listings, and figures.
- Cross-check endpoints against predefined objectives.
- Verify consistency between efficacy results and statistical outputs.
- Draft the CSR section using structured regulatory language.
- Flag discrepancies or missing data.
- Prepare a version ready for medical and regulatory review.
It is the same request, but very different level of execution. The first one writes text, the AI agent manages the process.
Why AI agents are especially relevant in pharma
Life sciences environments are complex by design. Every task often includes:
- Multiple data sources.
- Regulatory constraints.
- Cross-functional input.
- Audit requirements.
- Iterative validation steps.
This complexity makes agentic systems particularly valuable because they combine reasoning with structured tool usage. For example, in regulatory affairs, an AI agent-based solution can retrieve prior submissions, extract protocol data, cross-check endpoints against guidelines, draft sections, and highlight inconsistencies for human review.
These are not single prompts. They are sequences of controlled actions.
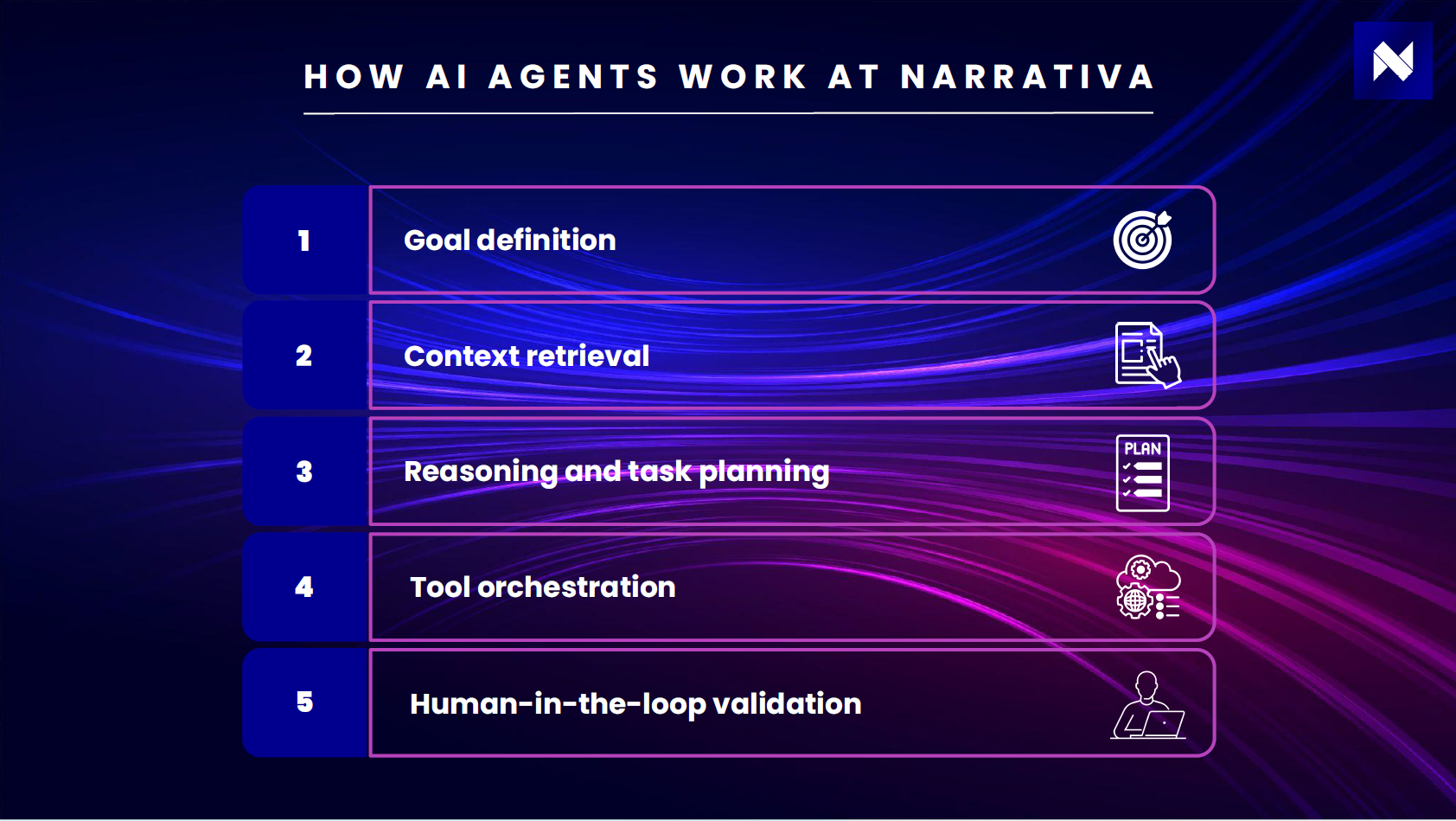
How AI agents work at Narrativa
At Narrativa, we don’t deploy generic AI agents. We design AI agent-based solutions for regulated environments. Every agent we build includes five core components:
1. Goal definition
The user defines a structured objective, such as drafting a clinical section or analyzing safety data.
2. Context retrieval
The agent gathers relevant documents, structured datasets, SOPs, and prior outputs.
3. Reasoning and task planning
It breaks the objective into smaller, traceable steps.
4. Tool orchestration
It interacts with internal systems such as:
- Clinical databases
- Document management platforms
- Knowledge repositories
- Internal API
5. Human-in-the-loop validation
All outputs remain reviewable, auditable, and version-controlled. This architecture ensures that autonomy operates within defined guardrails.
At Narrativa, we focus on building AI agents that operate inside clinical, regulatory, and operational environments with scientific rigor and compliance awareness. And in life sciences, where every step must be traceable, validated, and defensible, that operational intelligence makes all the difference.
So, are AI agents better than traditional AI?
Not always. It depends on the task. For simple activities such as basic summaries, short drafts, straightforward translations, a traditional model is often enough. But when a task requires:
- Multi-step reasoning
- Data retrieval from multiple systems
- Structured documentation
- Validation against rules
- Context persistence
An AI agent-based solution becomes the better solution, as most high-value pharma workflows fall into this second category. In short, they transform regulatory operations from fragmented, manual processes into coordinated, traceable, and continuously updated workflows.
About Narrativa
Narrativa® Agentic AI solutions unlock a faster, smarter future for life sciences organizations, helping them to efficiently produce complex, high-volume documentation for regulatory and commercialization workflows. By automating content creation, Narrativa® delivers greater speed, accuracy, and consistency—while ensuring full compliance in highly regulated environments.
The Narrativa® Navigator platform provides secure and specialized Agentic AI-powered automation features. It includes complementary user-friendly tools such as Clinical Atlas for CSR and Protocol generation, Narrative Pathway, TLF Voyager, and Redaction Scout, which operate cohesively to transform clinical data into submission-ready documents for regulatory and commercialization. From database to delivery, pharmaceutical sponsors, biotech firms, and contract research organizations (CROs) rely on Narrativa® to streamline workflows, decrease costs, and reduce time-to-market across the clinical lifecycle and, more broadly, throughout their entire businesses.
Explore www.narrativa.com and follow on LinkedIn, Facebook, Instagram, and X.

